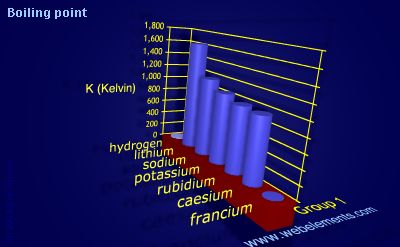
Potassium Properties and uses of Potassium The boiling point for potassium is 1425°F Potassium has the melting point of 146°F A slivery color is. - ppt download

Calculation the boiling point of a 1M aqueous solution (density 1.04 g mL^-1 )of potassium chloride (Kb for water = 0.52 K kg mol^-1 , Atomic masses: K = 39u, Cl =
Potassium. Kalium. Alkali metals. Chemical Element of Mendeleev's Periodic Table. Potassium in square cube creative concept Stock Photo - Alamy

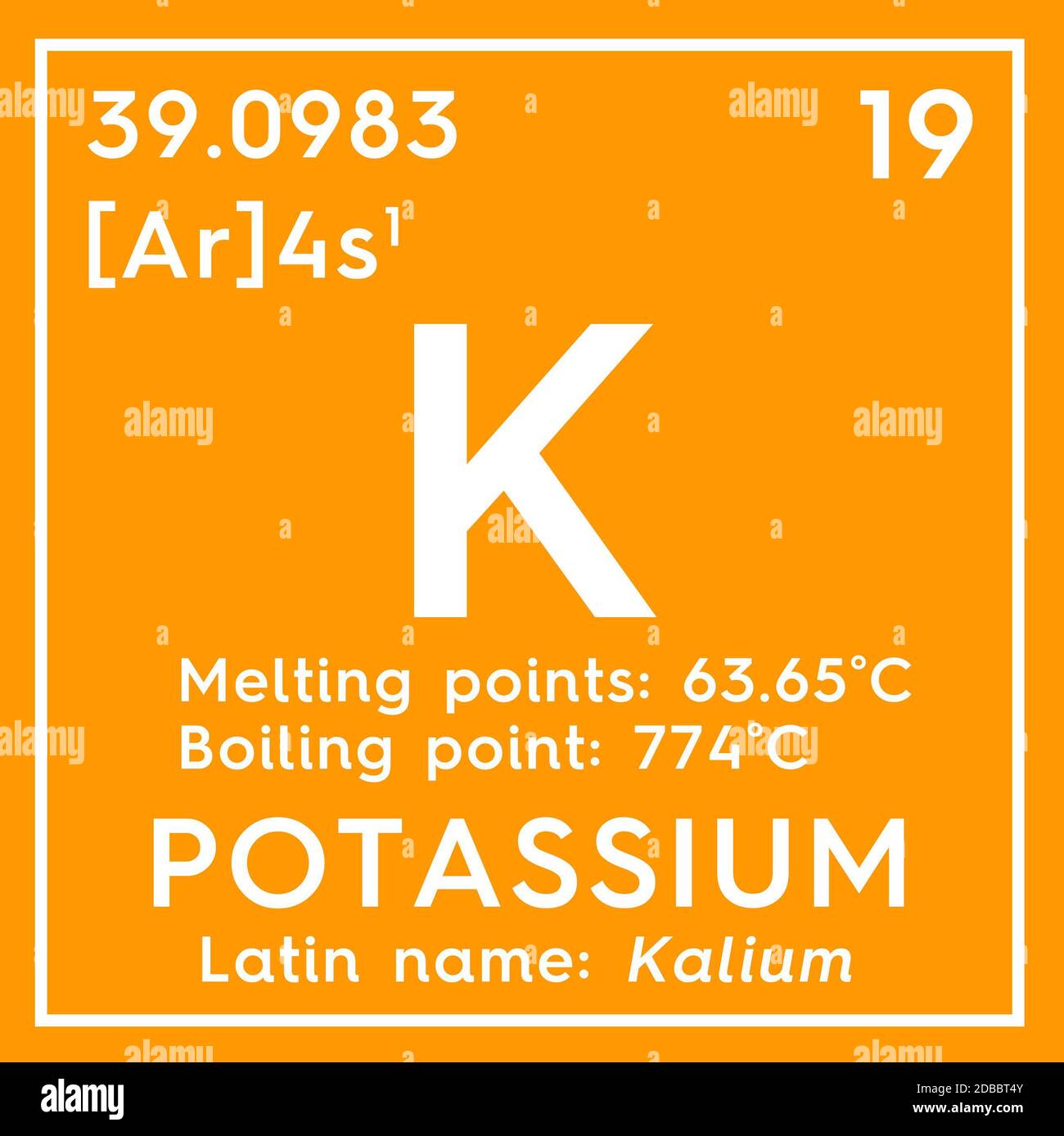
Chemistry Romania - >>Potassium< Group 1 Melting point 63.5°C, 146.3°F, 336.7 K Period 4 Boiling point 759°C, 1398°F, 1032 K Block s Density (g cm−3) 0.89 Atomic number 19 Relative atomic mass

Potassium Vector Illustration. Chemical Element Substance Characteristics Uses. Boiling Or Melting Temperature Diagram. Alkali Metals Part And Ionic Salts Ingredient. Educational Labeled Infographic Royalty Free SVG, Cliparts, Vectors, And Stock ...

Potassium Properties and uses of Potassium The boiling point for potassium is 1425°F Potassium has the melting point of 146°F A slivery color is. - ppt download

SOLVED: The boiling point of water is 100.0°C at atmosphere. How many grams of potassium nitrate (101.1 g/mol), must be dissolved in 288.0 grams of water to raise the boiling point by